To make this happen, the industry relies on ICH Q1A(R2) is the global gold standard for stability testing, providing a harmonized framework for the US, EU, and Japan to determine shelf life and storage conditions. Without these rules, companies would have to run different tests for every single country they sell in, which would be a logistical nightmare and incredibly expensive.
The Three Core Testing Categories
Stability testing isn't just one long experiment. It's split into three distinct tracks to give regulators a full picture of how a drug behaves under different levels of stress. You can't just pick one; you usually need data from all three to get a product approved.
- Long-term Testing: This is the "real world" simulation. It tells you how long the drug lasts under normal storage. For most markets, this means 25°C ± 2°C / 60% RH ± 5% RH or 30°C ± 2°C / 65% RH ± 5% RH. You typically need at least 12 months of this data before submitting your application to the FDA.
- Accelerated Testing: This is the "stress test." By pushing the drug to Accelerated Stability Testing conditions of 40°C ± 2°C and 75% RH ± 5% RH for 6 months, researchers can predict long-term degradation much faster. If a drug fails here, it's a huge red flag.
- Intermediate Testing: This is your safety net. You only trigger this (usually at 30°C ± 2°C / 65% RH ± 5% RH for 6 months) if the drug shows a "significant change" during the accelerated phase. It helps find the exact point where the product starts to break down.
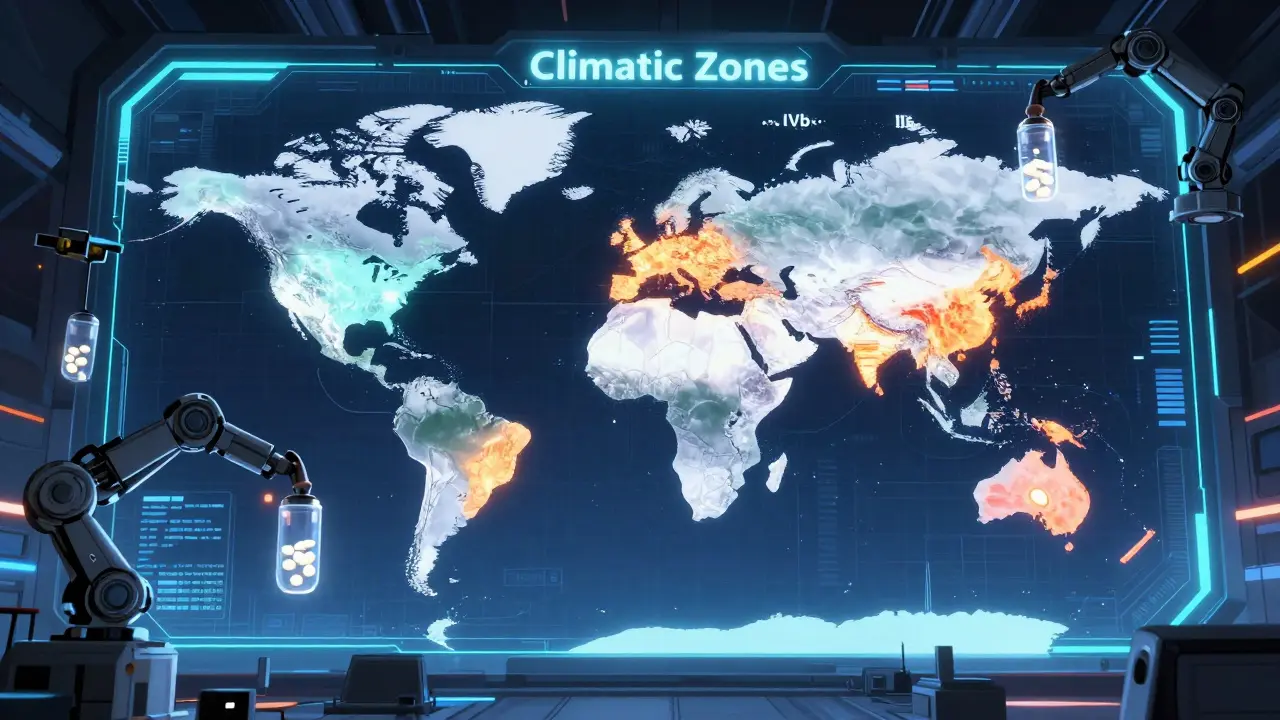
Mapping the Global Climatic Zones
Not all parts of the world are the same. A drug that stays stable in chilly Norway might degrade in the humid heat of Brazil. Because of this, Climatic Zones are geographical regions classified by temperature and humidity levels to ensure drug stability across different global environments. If you're targeting a global market, you have to tailor your long-term testing to the specific zone where the drug will be sold.
| Zone | Climate Type | Temperature | Relative Humidity (RH) |
|---|---|---|---|
| Zone I | Temperate | 21°C | 45% RH |
| Zone II | Mediterranean/Subtropical | 25°C | 60% RH |
| Zone III | Hot-Dry | 30°C | 35% RH |
| Zone IVa | Hot-Humid/Tropical | 30°C | 65% RH |
| Zone IVb | Hot/Higher Humidity | 30°C | 75% RH |
Time Intervals and the Testing Schedule
You can't just put a drug in a chamber and check it once a year. Stability is about the trend. Regulators want to see a clear trajectory of how the drug degrades over time. The standard schedule usually follows these intervals: 0, 3, 6, 9, 12, 18, 24, and 36 months.
If you expect a drug to be volatile, you'll need to test more frequently in the early stages. For example, if you're working with a new formulation that might degrade quickly, adding a 1-month or 2-month check can save you from a total study failure later on. The precision here is critical-environmental chambers must keep temperature within ±0.5°C and humidity within ±2% RH. A tiny drift in these numbers can invalidate months of work.
Special Considerations for Refrigerated Products
When you're dealing with biologics or temperature-sensitive vaccines, the standard 25°C or 40°C rules don't apply. For products that need refrigeration, Refrigerated Stability Testing protocols requiring long-term storage at 5°C ± 3°C for 12 months and accelerated conditions at 25°C ± 2°C / 60% RH for 6 months are used.
This is where things get tricky. Modern delivery systems, like the lipid nanoparticles used in mRNA vaccines, are incredibly sensitive. Standard ICH protocols often miss the damage caused by freeze-thaw cycles, which can cause irreversible degradation. This is why many companies are moving toward more dynamic testing rather than just static temperature points.

Common Pitfalls and Real-World Failures
In a perfect world, everything stays at 25°C. In the real world, power outages happen and sensors fail. Industry data shows that nearly 78% of professionals have dealt with a temperature excursion exceeding ±2°C during a study. When this happens, you can't just ignore it; you have to document the excursion and prove whether or not it affected the data.
Another major headache is the "significant change" threshold. The guidelines aren't always crystal clear on exactly how much degradation counts as a failure. This subjectivity can lead to fierce arguments with regulators. Imagine a scenario where a drug's assay result is 4.8% off the specification-statistically, it might be insignificant, but to a strict regulator, it could be the reason your application gets rejected.
The Future: From Physical Testing to Predictive Modeling
Waiting 36 months for a stability study to finish is a huge bottleneck for getting drugs to patients. To solve this, the industry is shifting toward Accelerated Predictive Stability (APS) a methodology using extremely high temperatures (50-80°C) and varying humidity to predict long-term stability in a fraction of the time.
While some regulators are still skeptical, about 74% of the top 20 pharma companies are already using these models. The goal is a risk-based approach where 60% of stability data is generated through modeling by 2030. We're also seeing a push toward Process Analytical Technology (PAT), which allows for real-time stability assessment during the manufacturing process itself, potentially cutting testing duration by 30-50%.
What happens if a drug fails accelerated stability testing?
If a "significant change" occurs during accelerated testing (40°C/75% RH), the company must conduct intermediate testing at 30°C/65% RH. This helps determine if the degradation was due to extreme stress or if it will likely happen under normal storage conditions. If it fails at the intermediate level too, the company must either change the formulation, the packaging, or shorten the proposed shelf life.
How long does long-term stability data need to be at submission?
For the FDA, the general requirement is 12 months of long-term data at the time of submission. However, the EMA (European Medicines Agency) offers more flexibility, sometimes allowing 6 months of data depending on the submission pathway chosen. This discrepancy is why global companies often default to the 12-month standard to satisfy all jurisdictions simultaneously.
What is a "significant change" in stability testing?
A significant change is typically defined as a 5% change in assay from its initial value, any degradation product exceeding its acceptance criterion, or a failure to meet specifications for appearance, functionality, or dissolution. Because some of these metrics can be subjective, it is often a point of contention during regulatory audits.
Why are there different climatic zones?
Climatic zones account for the fact that environmental stress varies by geography. For example, a product sold in Zone IVb (Hot/Higher Humidity) faces much higher moisture stress than one in Zone I (Temperate). Testing against these specific zones ensures that a drug remains stable regardless of where in the world it is stored.
Can predictive modeling replace real-time stability testing?
Not entirely. While Accelerated Predictive Stability (APS) and other models are increasingly used to speed up development and screen formulations, regulators still require real-time, long-term data to finalize the shelf life and storage labels. Models act as a guide, but the physical data is the legal proof.




Anastasios Kyriacou
April 22, 2026 AT 09:31too long didnt read just use aps and get it over with lol
Divyanshu Giri
April 23, 2026 AT 12:45This is a total goldmine of info!! Keep pushing the boundaries of science and let's make the world healthier together!! Super power stuff!! 🚀
Rick Brewster
April 24, 2026 AT 18:10one must ponder the inherent fragility of human existence mirrored in the decay of a molecule where we seek to impose a rigid order through these ich guidelines but really we are just dancing with the inevitability of entropy and the fda is merely the choreographer of our collective fear of the unknown which is frankly quite quaint when you consider the vastness of cosmic time and the sheer futility of wanting a pill to last exactly three years in a humid basement in brazil
Ally Warren
April 25, 2026 AT 04:53There is a certain irony in our attempt to quantify stability in an unstable world. We build these precise chambers to simulate a reality that is fundamentally chaotic, hoping that by controlling the humidity, we can somehow outrun the clock of degradation.
RAJESH MARAVI
April 26, 2026 AT 05:24Absolute joke... who even cares about Zone I when the real world is failing?? The data is probably faked anyway and these "predicitive models" are just fancy ways for pharma to steal more money from patients while the drug degrades in the box. pathetic lol
Caroline Duvoe
April 27, 2026 AT 08:17everyone knows the 5% assay rule is basically arbitrary 🙄 the industry has been coasting on these standards for decades and it's honestly embarrassing that we're still talking about it like it's new info
Saptatshi Biswas
April 29, 2026 AT 05:03The audacity to suggest that European standards are "flexible" while the US is "strict" is simply laughable. Our laboratories in India are far more rigorous than these mediocre Western facilities, and we manage the most complex climatic zones in the world with far superior precision. Your analysis fails to acknowledge the sheer scale of Indian pharmaceutical dominance!
Amy Fredericks
April 30, 2026 AT 06:17I think it's wonderful that we're seeing a shift toward predictive modeling. It really opens the door for faster drug delivery to people who need it most, and I'm sure the regulators will come around once the data proves it's safe. Let's stay positive about these innovations!
Olayinka Ibukunoluwa Mercy
May 1, 2026 AT 11:17I completely agree with the need for a more inclusive approach to global health!! 🌍 It is so important that we ensure medications are stable for everyone, regardless of where they live... especially in tropical regions where humidity is a real challenge!! Keep up the great work, everyone!! ✨🙏🌟